| |
|
| Mutinaite |
|Na3 Ca4
(H2O)60| [Al11Si85O192]
|
| |
|
|
|
| Morphology: |
|
|
| |
Subspherical aggregates
(up to 1.8 mm) of tiny radiating lath-like fibers or as aggregates
of transparent tabular crystals. |
|
| |
|
| Physical
properties: |
|
| |
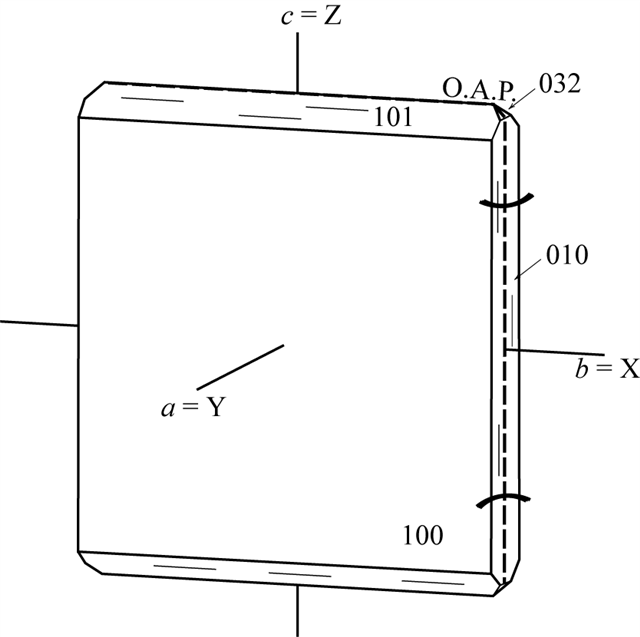
Cleavage: {100} good.
Hardness: 3 - 4.
Density: 2.14 (obs) gm/cm3.
Luster: vitreous.
Streak: white. |
|
| |
| Optical
properties: |
| |
Color: colorless to white;
colorless in thin section.
Biaxial (-). α = 1.485, β= 1.487, γ = 1.488, δ = 0.003, 2Vx =
70°.
O.A.P || (100). |
| |
| Crystallography: |
| |
Unit cell
data:
a 20.201, b 19.991, c 13.469 Å, Z
= 1, Space Group Pnma (Vezzalini et al.
1997). |
| |
| |
| |
|
|
| Name: |
|
| |
Mutinaite was named and
described by Galli et al. (1997). The type locality is in
the Ferrar dolerite, exposed in the southwest crest, just under Mt.
Adamson, Antartica. The name is for Mutina, the ancient Latin name
of Modena, Italy, recognizing the Department of Earth Science at the
University of Modena as an active center for zeolite research. |
| |
|
|
|
| Crystal structure: |
|
| |
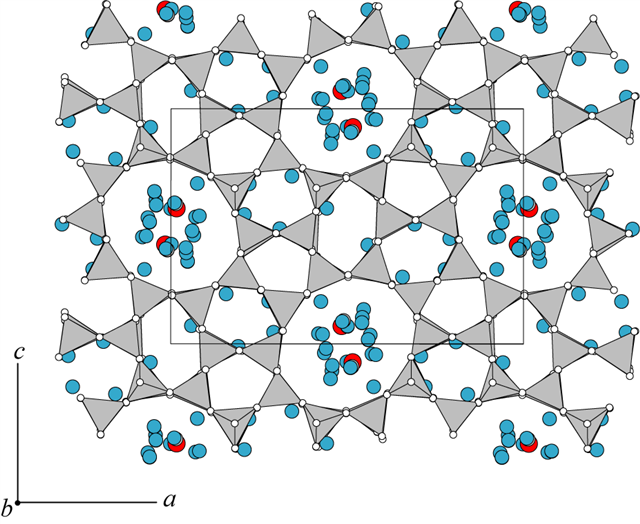
The framework topology (MFI)
of mutinaite (Vezzalini et al. 1997) is the same as that
of the synthetic zeolite ZSM-5 (Olson et al. 1981).
Mutinaite, however, has a much higher content of Al than any of the
ZSM-5 frameworks, for which the structure has been refined. The
pentasil nature of the framework can be seen in a sheet parallel to
010 (accompanying figure). The building units of this sheet are the
mfi (5454), cas (5262),
and tes (54) polyhedra. |
| |
These units, linked by
sharing of edges or faces, form 10-membered rings. The resulting
framework contains two intersecting channels, one parallel to [010]
form the 10-ring openings of the 010 sheet, and the other is
sinusoidally parallel to [100]. The effective diameters of the
10-rings of the [010] channel are 6.1 x 4.6 Å, and that of the
10-rings of the [100] sinusoidal channel, about 5.1 Å.
The narrow range of T-O distances, 1.587-1.618 Å, is compatible with
a disordered Si-Al distribution (Vezzalini et al. 1997).
Although 17 non-framework sites were located within the channels all
have such low electron densities that partial cation occupancy could
not be distinguished from H2O molecules. One site,
tentatively assigned to Ca2+ (red), is not coordinated
with any framework oxygen.
The dehydration behavior of the synthetic counterpart of mutinaite,
zeolite ZSM-5, was investigated by van Koningsveld (1990) and Precisvalle
et al. (2023). The structural deformations induced by pressure in mutinaite
were studied by means of complete Rietveld structural refinements
(Quartieri et al. 2012).
|
 |
| |
|
| Chemical composition: |
| |
The only analysis for
mutinaite is given by (Galli et al. 1997). TSi
value of 0.883 is the highest of any known natural zeolite. All
synthetic analogs of multinaite (ZSM-5 and silicalite) have even
higher silica contents. |
| |
|
| Occurrences: |
| |
Mutinaite has been found
in only one occurrence, vesicles and cavities in the Jurassic Ferrar
dolerite of Mt. Adamson, Northern Victoria Land, Antarctica. It is
associated with other zeolites, mordenite, heulandite, erionite,
phillipsite, stilbite, levyne, epistilbite, tschernichite, boggsite,
gottardiite, ferrierite, and cowlesite, as well as quartz,
cristobalite, apophyllite, gypsum, and calcite. Galli et al.
(1997) suggest that the rare, disordered, high-silica zeolites in
this association must have formed under special conditions, such as
rapid environmental cooling during crystal growth. |
| |
|
| References: |
| |
Galli, E., Vezzalini, G.,
Quartieri, S., Alberti, A., and Franzini, M. (1997) Mutinaite, a new
zeolite from Antarctica: the natural counterpart of ZSM-5. Zeolites.
19, 318-322.
Olson, D.H., Kokotailo, G.T. & Lawton, S.L. (1981) Crystal
structure and structure-related properties of ZSM-5. J. Phys.
Chem. 85, 2238-2243.
Precisvalle, N., Mancinelli, M., Ardit, M., Beltrami, G., Gigli, L., Aloise, A.,
Catizzone, E., Migliori, M., Giordano, G., Guidi, V. and Martucci, A. (2023)
Temperature Induced Monoclinic to Orthorhombic Phase Transition in Protonated
ZSM-5 Zeolites with Different Si/Al Ratios: An In-Situ Synchrotron X-ray Powder
Diffraction Study. Crystals, 13(6), 979.
Quartieri, S., Arletti, R., Vezzalini, G., Di Renzo, F. and Dmitriev, V. (2012)
Elastic behavior of MFI-type zeolites: 3–Compressibility of silicalite and
mutinaite. J. Solid State Chem., 191, 201-212.
Van Koningsveld, H. (1990) High-temperature (350 K) Orthorhombic
Framework Structure of Zeolite H-ZSM-5. Acta Cryst. B, 46, 731–735.
Vezzalini, G., Quartieri, S., Galli, E., Alberti, A., Cruciani, G.,
and Kvik, Å. (1997) Crystal structure of the zeolite mutinaite, the
natural analog of ZSM-5. Zeolites. 19,
323-325.
Updated: October 2025.
|