
Cowlesite on saponite-chlorite, Yacolt, Clark County, Washington, USA
Occurs as hemispheres of delicate, very thin blades
Common forms: {010}, {101} and {100}
Hardness: 5 - 5½
Density: 2.14(2) g/cm3
Luster: pearly
Streak: white
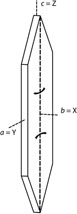
Biaxial (-)
α = 1.505-1.513, β = 1.509-1.516,
γ = 1.509-1.518, δ = 0.005
2Vx = 30-53°
X = b, Y = a, Z = c
O.A.P. || (010)
Dispersion: none
b 24.891 Å
c 30.468 Å
 The extreme thinness of the crystals and stacking of plates have
prevented a determination of the structure for a very long time.
Recently, ab initio structure determination of this mineral was
obtained by three-dimensional electron diffraction from
single-crystal domains of a few hundreds of nanometers (Mugnaioli et
al. 2020). The structure of cowlesite consists of an
alternation of rigid zeolitic layers and low-density interlayers
supported by water and cations. This makes cowlesite the only
two-dimensional zeolite known in nature. The structure of a single
layer can be decomposed in just two independent building units
(CBU), a d6r (12T) and a heavily distorted imf
(16T), the latter never observed in natural zeolites. Two additional
T atoms are connected with both d6r and imf and
are responsible for the interlayer Si–O–Si bridging (Mugnaioli et
al. 2020).
The extreme thinness of the crystals and stacking of plates have
prevented a determination of the structure for a very long time.
Recently, ab initio structure determination of this mineral was
obtained by three-dimensional electron diffraction from
single-crystal domains of a few hundreds of nanometers (Mugnaioli et
al. 2020). The structure of cowlesite consists of an
alternation of rigid zeolitic layers and low-density interlayers
supported by water and cations. This makes cowlesite the only
two-dimensional zeolite known in nature. The structure of a single
layer can be decomposed in just two independent building units
(CBU), a d6r (12T) and a heavily distorted imf
(16T), the latter never observed in natural zeolites. Two additional
T atoms are connected with both d6r and imf and
are responsible for the interlayer Si–O–Si bridging (Mugnaioli et
al. 2020). When cowlesite gets in contact with a transmission electron microscope vacuum, a phase transition to a conventional 3D zeolite framework occurs in a few seconds. The original cowlesite structure could be preserved only by adopting a cryo-plunging sample preparation protocol. Continuous loss of loosely bonded water from cowlesite interlayer destabilizes the Ca-rich interlayer and triggers the phase transition toward a 3D zeolitic framework around 411 K at ambient pressure (Mugnaioli et al. 2020).